BEHAVIORAL NEUROPHARMACOLOGY LAB’S
PRECLINICAL MODELS
Our approach to understanding the processes mediating the addictive effects of drugs requires the use of many different behavioral tasks. This multi-task approach is necessitated by the complex action of drugs in the central nervous system (CNS). From our perspective, one way to understand this complex action is to take advantage of different tasks that have been developed to understand different components of a drug’s action. For example, self-administration is well suited to measure the reinforcing effects of a drug, whereas conditioned taste avoidance measures the aversive- or avoidance-inducing effects. Although some of our research uses these tasks as mere assays, you will notice that much of our research is designed to understand the functional relations (behavioral and neuropharmacological) that affect what is being learned.
Locomotor Activity

Drugs of abuse, as well as current and potential pharmacotherapies, often affect the general activity of rats. For example, cocaine, amphetamine, methamphetamine, and bupropion (Zyban®) are potent stimulants that increase rats’ general activity. Interestingly, nicotine has a biphasic effect on rats’ activity. Early administrations often suppress locomotor activity, whereas later administrations tend to have a stimulant effect. We assess the unconditioned and conditioned locomotor effects of many compounds in our laboratory using circular chambers with infrared emitter/detector units interfaced with a computer [Nb. we will be moving to video tracking soon]. Our laboratory has been especially interested in the locomotor conditioning effects of nicotine. Conditioning refers to the observation that a distinct environment (i.e., locomotor chambers) will come to control an increase in activity, in the absence of any drug, if the rat receives the environment repeatedly paired with the locomotor stimulant effects of the drug. This control of activity by an environment is thought to reflect a Pavlovian conditioning process in which the environment is the conditional stimulus (CS) and the locomotor effects of the drug serve as the unconditional stimulus (US). The increase in activity evoked by the environmental CS is the conditioned response (CR). Many theories of drug abuse implicate Pavlovian conditioning in the addiction process. A subset of these theories places emphasis on locomotor sensitization (i.e., enhanced sensitivity to drug with repeated exposure) and the underlying conditioned and unconditioned processes. Accordingly, understanding the behavioral and neurobiological factors responsible for the locomotor effects of drugs could provide important insight into drug abuse processes and help identify potential interventions.
Place Conditioning

As noted earlier, stimuli that occur in close temporal and spatial proximity to the CNS effect of an abused drug can enter into a conditioned association with these drug effects. Notably, these associations can impact choice behavior. The place conditioning procedure is ideally suited to study how these conditioned associations alter choice in a rodent model. In a typical place conditioning experiment in our laboratory, rats receive one distinct environment paired with a drug or some other stimulus (e.g., novel object—others have studied food, sucrose, copulatory opportunity, wheel running, etc.). In a distinctly different environment, the rat receives nothing. This protocol is usually repeated for 4 to 8 exposures to each environment. On the test day, rats in a drug-free state are placed in the chamber and given free access to both environments (i.e., a choice test). If the drug (or other stimulus) has rewarding properties then the rat spends more time in the paired compartment. This result is often termed a conditioned place preference and is thought to result from the environmental cues acquiring appetitive properties (conditioned reward). Stimuli that have a conditioned or unconditioned rewarding quality tend to control approach-like behaviors. This conditioned-approach tendency presumably increases the amount of time in the drug-paired environment. Notably, the place conditioning procedure can also detect whether a drug or stimulus has an aversive quality. That is, rats tend to avoid the environment that was paired with an aversive stimulus (e.g., foot-shock, illness). We have used this model to study the parallel effects between novelty reward and drug reward. This interest is based on the observation that individuals classified as high novelty/sensation seekers tend to engage in more risky behaviors—including drug taking.
Self-administration
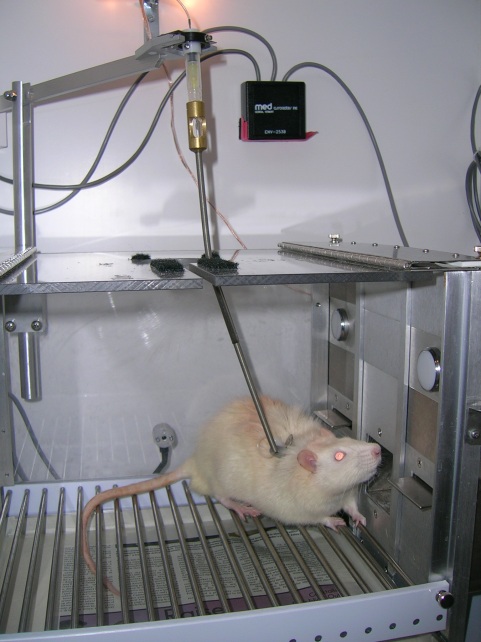
An important assumption of many drug addiction theories is that abused drugs have a reinforcing effect. This reinforcing effect, in part, is responsible for acquisition and maintenance of drug-taking behavior. If this assumption is correct, a preclinical model should be able to take an abused drug and use it to shape and maintain a behavior. The drug self-administration task provides support for this assumption and is arguably the most face valid model of human drug taking. In this task, rats are surgically prepared with an intravenous (IV) catheter. Then, in an operant conditioning chamber two response options (left lever press versus right lever press) are made available to the rat. One response is associated with IV infusion of a drug; the other response has no consequence. Most abused drugs differentially increase the rate of responding on the drug-associated lever. This task serves as an important preclinical model for the effectiveness of pharmacotherapies and immunotherapies to decrease drug-taking behavior. Moreover, this model can be used to study the importance of the response-reinforcer relation as well as the impact of stimuli present during self-administration. Recently, there has been a significant increase in interest in the role of these stimuli in the maintenance and re-instatement of self-administration.
Drug Discrimination (Interoceptive Conditioning)
The pharmacological effects of a drug can have distinct interoceptive cueing effects. Take as an example the light-headedness, warmth, etc. associated with having a few beers or the jitteriness and increase heart rate associate with drinking several cups of coffee. These cueing effects can play an important role in the addiction process such as prompting additional drug seeking.
Operant conditioning protocol. The standard preclinical rodent model for studying the cueing (discriminative stimulus) effects of a drug is referred to as operant drug discrimination. In this task, there are typically two response options available in the chamber. When the rat is pretreated with a drug, one response option (left lever press) is food reinforced under some schedule [e.g., fixed ratio 25 (FR25)—25th press produces food]. The other response option (right lever press) is not reinforced. When the rat is treated with saline (no drug) the response-reinforcer relation is switched. That is, right lever presses are reinforced, whereas left lever presses have no consequence. Rats (like humans) can readily learn to discriminate between a drug versus no drug state pressing the food-associated lever over 90% of the time.
Pavlovian conditioning protocol.
Although we have done some research with the operant conditioning protocol, much of our current empirical effort is focused on studying drug cues from a Pavlovian conditioning perspective (i.e., interoceptive conditioning). We have been studying two variants of this task. In one variant, the drug stimulus (e.g., nicotine) signals intermittent access to a sucrose reward; saline signals the absence of the reward. In this example, nicotine readily comes to control anticipatory sucrose-seeking behavior relative to saline (i.e., a goal tracking CR). Nicotine is conceptualized as a contextual cue (CS) that enters into a conditioned association with the sucrose US. Current research has supported this assumption. In the second variant, the drug state signals that a brief stimulus (e.g., 15-sec light) will be followed by sucrose. When saline is administered, the same light will not be followed by sucrose. Thus, the drug state signals when the light CS-sucrose US relation is in effect. Rats readily learn this discrimination showing an increase in goal tracking in the drug state only during the CS. From a Pavlovian conditioning perspective the drug stimulus in this task serves as an occasion setter or a feature positive modulator.
Other Models
Conditioned taste avoidance. There is a seeming paradox with abused drugs. Although approach, reward, reinforcement are among their central nervous system effects, drugs of abuse can also condition avoidance tendencies. There is still debate as to whether this avoidance reflects an aversive quality to the drug or some other effect. Regardless, this effect and its neurobiological processes can be studied using a preparation termed conditioned taste avoidance (aversion). In this task, consumption of a novel taste such as saccharin (i.e., the CS) is followed by administration of the drug (i.e., the US). Relative to controls, rats drink less of the drug-paired taste.
Drug withdrawal. Cessation from chronic drug use is associated with a wide array of behavioral and neurobiological effects that can prompt relapse. To study drug withdrawal, rats have an osmotic pump loaded with the drug of interest (e.g., nicotine) surgically implanted subcutaneously. The osmotic pump releases a relatively consistent amount of drug across time. Withdrawal is precipitated by removal of the pump or administration of an antagonist. For example, mecamylamine and naloxone can induce nicotine withdrawal signs in rats receiving chronic nicotine.
Object recognition (novelty detection). Rats can readily detect novelty in the environment. The object recognition task takes advantage of this tendency to measure memory and to test manipulations (pharmacological, neurobiological, and behavioral) that improve or impair memory. In this task, rats are familiarized with an object. Some time later, the rat is placed in an environment with the familiar object and a novel object. Rats spend more time interacting with the novel object indicating recall of the familiar object. Long delays to testing or familiarization in a different environment than the test environment can impair object recognition. Drugs found to improve memory in Alzheimer’s patients also improve deficits in object recognition.
Environment familiarization. Almost every task used to study drug effects and learning processes involves exposure to a test environment. During exposure to the environment the rat learns about the stimulus features of the environment. This learning (i.e., environmental familiarization) can affect ongoing behavior in a task. Accordingly, a manipulation that affects familiarization could produce a nonspecific change in the behavior of interest. If so, an experimenter could mistakenly attribute a behavior change to the wrong cause. For this reason, factors affecting environmental familiarization are of much interest. We measure environmental familiarization by taking advantage of a rat’s tendency to interact more with a novel object in a familiar environment than in a novel environment.